A recent exploratory clinical study published in the Journal of Clinical and Experimental Dentistry investigated the use of high-sensitivity C-reactive protein (hs-CRP) levels in pulpal blood as a potential objective biomarker for assessing the severity of irreversible pulpitis.
Traditionally, the diagnosis of irreversible pulpitis relies heavily on subjective pain reports and clinical tests such as thermal stimulation, which may vary between patients and practitioners. The study explored whether quantitative measurement of hs-CRP directly from pulpal blood could provide a more objective indicator of inflammatory status.
In this study, hs-CRP levels were measured using the Finecare™ FIA Meter Plus. With compact design, extended reagent shelf life, and user-friendly operation, Finecare™ FIA Meter series is well suited for decentralized settings such as small clinics and specialty practices where timely quantitative testing is essential.
Key Findings
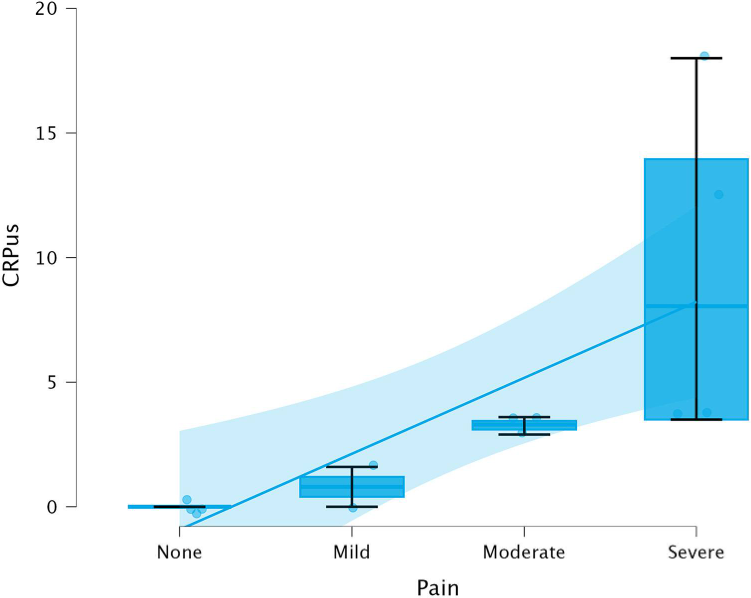
hs-CRP levels demonstrated a strong positive correlation with patient-reported pain intensity.
Pulpal blood hs-CRP levels increased with pain severity, showing a strong correlation with inflammatory status (ρ = 0.918).
Source: Muñoz P. et al., Frontiers in Dental Medicine, 2026 (CC BY 4.0).
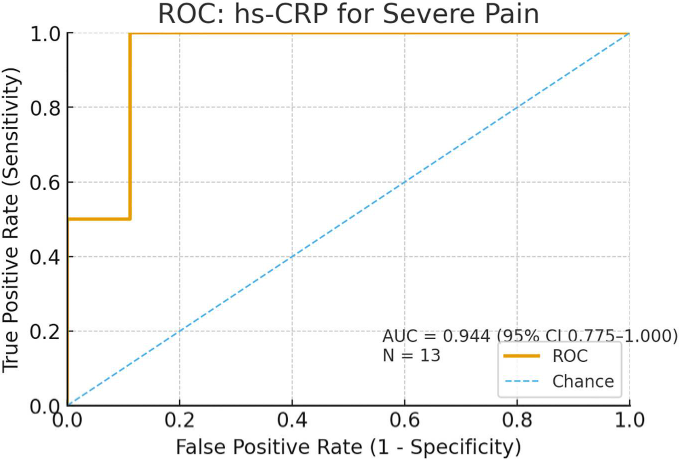
A threshold value of ≥ 3.4 mg/L showed high sensitivity and specificity in distinguishing more severe inflammatory cases within the study population.
The authors suggested that hs-CRP measurement in pulpal blood may serve as a promising adjunctive biomarker for inflammation severity assessment.
High-sensitivity CRP demonstrated excellent diagnostic performance for severe pulpal inflammation (AUC = 0.944).
Source: Muñoz P. et al. Rapid point-of-care quantification of hs-CRP in pulpal blood for diagnosing irreversible pulpitis. Frontiers in Dental Medicine, 2026. Licensed under CC BY 4.0.
Although the study was conducted as a pilot investigation with a limited sample size, the findings introduce a novel application perspective for hs-CRP testing in dental clinical settings.
Expanding the Scope of Point-of-Care Inflammation Testing
C-reactive protein is widely recognized as a systemic inflammation marker in cardiovascular and infectious disease management. This study presents an innovative cross-disciplinary application, exploring its potential value in localized dental inflammation.
The use of the Finecare™ FIA Meter Plus in this research highlights the adaptability of rapid fluorescence immunoassay technology in diverse clinical scenarios, including emerging point-of-care applications beyond traditional laboratory settings.
Wondfo remains committed to supporting clinical research and expanding the potential applications of rapid quantitative immunoassay technologies across multiple medical specialties.
Disclaimer:
The referenced study represents exploratory research findings and further large-scale clinical investigations are required to validate its clinical applicability.
Reference:
Front Dent Med. 2026 Jan 7;6:1729860. doi: 10.3389/fdmed.2025.1729860
 The first developed technology platform with various application scenarios, including infectious disease, fertility, DOA, etc.
The first developed technology platform with various application scenarios, including infectious disease, fertility, DOA, etc. 50+ kinds of reagents and five high-performance devices, focusing on detecting cardiovascular disease, inflammation, kidney injury, sex hormones, thyroid function, diabetes, tumor, and others.
50+ kinds of reagents and five high-performance devices, focusing on detecting cardiovascular disease, inflammation, kidney injury, sex hormones, thyroid function, diabetes, tumor, and others. Single-dose Chemiluminescense Immunoassay Platform
Single-dose Chemiluminescense Immunoassay Platform Wondfo optical blood coagulation analyzer is the first one in the world that can test PT, APTT, TT, FIB, and ACT simultaneously.
Wondfo optical blood coagulation analyzer is the first one in the world that can test PT, APTT, TT, FIB, and ACT simultaneously.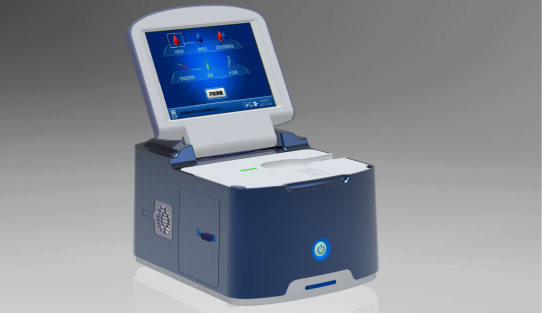 Our Blood Gas Analyzer BGA-102 can produce the result in 30s. Its advantages of portability, easy operation, durability, and high performance make it ideal for clinics, laboratories, and hospitals.
Our Blood Gas Analyzer BGA-102 can produce the result in 30s. Its advantages of portability, easy operation, durability, and high performance make it ideal for clinics, laboratories, and hospitals. Ready-to-use lyophilized RT-PCR Reagent;
Ready-to-use lyophilized RT-PCR Reagent; Wondfo PA-3600 IHC Staining System
Wondfo PA-3600 IHC Staining System The Wondfo Truth-H80E HPLC Hemoglobin Analyzer is a high-performance diagnostic device designed for rapid and precise measurement of glycosylated hemoglobin (HbA1c)—the gold standard for diabetes diagnosis.It support the standard mode and variant mode and provide the result within 60-90seconds.
The Wondfo Truth-H80E HPLC Hemoglobin Analyzer is a high-performance diagnostic device designed for rapid and precise measurement of glycosylated hemoglobin (HbA1c)—the gold standard for diabetes diagnosis.It support the standard mode and variant mode and provide the result within 60-90seconds. Finecare™ Vitamin D, from complexity to simplicity
Finecare™ Vitamin D, from complexity to simplicity Building A World Free from Antibitoic Overuse
Building A World Free from Antibitoic Overuse Advanced rapid diagnostic test with WHO prequalification for infectious disease
Advanced rapid diagnostic test with WHO prequalification for infectious disease The Future Intelligent Medical Assistant to Healthcare
The Future Intelligent Medical Assistant to Healthcare Fight against the pandemic through continuous innovation
Fight against the pandemic through continuous innovation