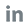100% Sensitivity, Another Recognition for Finecare™ FIA Meter
April 14,2023 (Edit) Wondfo
Recently, an evaluation of fluorescence immunoassay in quantitative measuring of hemoglobin A1c (HbA1c) in diabetes patients was published on Frontiers in Bioscience-Landmark. It verifies that Wondfo’s Finecare™ FIA HbA1c Test is a reliable and rapid assay which can be easily implemented for long-term monitoring of HbA1c in diabetic patients. The Finecare™ FIA Systems, as the first developed-by-China quantitative fluorescence immunoassay analyzers in 2009, feature a comprehensive test menu, support various sample types (whole blood/serum/plasma) and can detect several different items simultaneously. Currently, more than 70 thousand Finecare™ FIA Meters have been installed worldwide.

100% Sensitivity, Compared with Globally Top-Class System
Frontiers in Bioscience-Landmark (FBL) is an international peer-reviewed journal in the field of biological technology, dedicated to publishing advances in biology.
On March 23, 2023, an evaluation of fluorescence immunoassay in quantitative measuring of HbA1c was published on Frontiers in Bioscience-Landmark. Led by Qatar University, it analyzed a total of 100 samples (fingerstick and venipuncture whole blood) with Wondfo’s Finecare™ HbA1c Rapid Quantitative Test and the results were compared with the reference assay, a globally top-class biochemistry-immunoassay system.
The evaluation concludes that Finecare™ FIA HbA1c assay showed excellent agreement with the reference assay as the mean bias was negligible. A very small mean bias was shown between the fingerstick and the venipuncture data. Finecare™ showed 100% sensitivity and 98.7% specificity compared to the reference system using venipuncture samples.
Moreover, Wondfo’s Finecare™ showed a significant difference between normal, pre-diabetic and diabetic samples (p < 0.0001). According to the conclusion of the evaluation, Wondfo’s Finecare™ System is a reliable and rapid assay (5 min) which can be easily implemented for long-term monitoring of HbA1c in diabetic patients.
Performance Verified, We Aim to Make Technology Benefit Everyone
Since the launch of Finecare™ FIA Meters in 2009, the system has been widely distributed in 100+ countries and regions and highly appreciated by many users. Finecare™ System is easy to operate, rapid in delivering results (in 15 mins), capable of testing different items simultaneously and equipped with a comprehensive testing panel allowing various sample types (whole blood/serum/plasma). Up to now, more than 70 thousand units of Finecare™ FIA Meters have been installed in over 100 countries and regions worldwide, serving laboratories, ERs, ICUs, Cardiology Depts, etc.
Currently, Finecare™ FIA Meters provide a comprehensive testing panel with 50+ items, covering markers of cardiovascular disease, inflammation, cerebral injury, diabetes and renal disease, tumor, thyroid hormone, etc. With the most comprehensive panel, Finecare™ FIA Meters of different models (single-channel, semi-automatic and automatic) can be flexibly applied in various scenarios.

Besides HbA1c’s excellence verified by FBL, a lot of Finecare™ rapid assays have been verified in outstanding performance. In September 2022, Clinical Chemistry and Laboratory Medicine, an authoritative journal of laboratory medicine with an impact factor of 8.490, published a research on the point-of-care detection of TSH (thyroid stimulating hormone). The essay verifies that Wondfo fluorescence immunoassay system can effectively assist in the clinical diagnosis of thyroid dysfunction. In addition, in February 2023, Department of Virology Institute of Tropical Medicine Nagasaki University, a WHO collaborating centre, delivered an evaluation of Wondfo Finecare™ Dengue NS1 Antigen test kit. As the evaluations shows, Finecare™ Dengue NS1 Ag test kit (immunofluorescence) enjoys excellent performance and can be of great help in controlling dengue fever.
With continuous efforts in research and innovation, Wondfo successively improved Finecare™ System in technology, realizing whole-process automatic measurement of infection. Only one step operation is needed for WBC+IPCS. Automatic testing with minimal volume of whole blood sample is realized through direct sampling without decapping. It is able to be connected with hemocytometer through mechanical arm, reduce manual steps to load sample rack. Many more delicate designs aim to streamlines manual operation, such as 360° barcode scanning, bi-directional LIS, RFID label scanning, etc.

Based on Wondfo’s nine technology platforms and multiple product lines, Wondfo has launched many scenario-targeted solutions, including solutions for acute and critical cases, inflammation, coagulation, primary laboratory, chest pain center, pathology, etc.
Dedicated to make biology technology benefit the public, Wondfo strives for excellence with continuous efforts for the better, so as to improve the efficiency of diagnosis in medical institutions and facilitate the construction of a Healthy China.
 The first developed technology platform with various application scenarios, including infectious disease, fertility, DOA, etc.
The first developed technology platform with various application scenarios, including infectious disease, fertility, DOA, etc. 50+ kinds of reagents and five high-performance devices, focusing on detecting cardiovascular disease, inflammation, kidney injury, sex hormones, thyroid function, diabetes, tumor, and others.
50+ kinds of reagents and five high-performance devices, focusing on detecting cardiovascular disease, inflammation, kidney injury, sex hormones, thyroid function, diabetes, tumor, and others. Single-dose Chemiluminescense Immunoassay Platform
Single-dose Chemiluminescense Immunoassay Platform Wondfo optical blood coagulation analyzer is the first one in the world that can test PT, APTT, TT, FIB, and ACT simultaneously.
Wondfo optical blood coagulation analyzer is the first one in the world that can test PT, APTT, TT, FIB, and ACT simultaneously. Our Blood Gas Analyzer BGA-102 can produce the result in 30s. Its advantages of portability, easy operation, durability, and high performance make it ideal for clinics, laboratories, and hospitals.
Our Blood Gas Analyzer BGA-102 can produce the result in 30s. Its advantages of portability, easy operation, durability, and high performance make it ideal for clinics, laboratories, and hospitals. Ready-to-use lyophilized RT-PCR Reagent;
Ready-to-use lyophilized RT-PCR Reagent; Wondfo PA-3600 IHC Staining System
Wondfo PA-3600 IHC Staining System The Wondfo Truth-H80E HPLC Hemoglobin Analyzer is a high-performance diagnostic device designed for rapid and precise measurement of glycosylated hemoglobin (HbA1c)—the gold standard for diabetes diagnosis.It support the standard mode and variant mode and provide the result within 60-90seconds.
The Wondfo Truth-H80E HPLC Hemoglobin Analyzer is a high-performance diagnostic device designed for rapid and precise measurement of glycosylated hemoglobin (HbA1c)—the gold standard for diabetes diagnosis.It support the standard mode and variant mode and provide the result within 60-90seconds. Finecare™ Vitamin D, from complexity to simplicity
Finecare™ Vitamin D, from complexity to simplicity Building A World Free from Antibitoic Overuse
Building A World Free from Antibitoic Overuse Advanced rapid diagnostic test with WHO prequalification for infectious disease
Advanced rapid diagnostic test with WHO prequalification for infectious disease The Future Intelligent Medical Assistant to Healthcare
The Future Intelligent Medical Assistant to Healthcare Fight against the pandemic through continuous innovation
Fight against the pandemic through continuous innovation