On August 25, Wondfo Biotech released the 2022 mid-year report. In the first half of the year, we continued to focus on the main business and achieved rapid growth despite the high base last year. Operating income was 4.034 billion yuan, a year-on-year increase of 117.30%; net profit attributable to the parent was 1.178 billion yuan, a year-on-year increase of 111.98%; net cash flow from operating activities was 1.128 billion yuan, a year-on-year increase of 239.52%.
Fast Increase in Infectious Disease Testing
Breakthrough of CLIA Testing
In the first half of 2022, among four major businesses, the revenue from infectious disease testing reached 3.207 billion yuan, a sharp increase of 240.32% over the same period last year; the sales revenue of COVID-19 testing kits was about 3.020 billion yuan. Testing for chronic disease management realized revenue of about 512 million yuan. Slight declining was observed due to pandemic prevention and control, but the chemiluminescence immunoassay testing achieved breakthrough growth. The income from fertility testing achieved 127 million yuan, a year-on-year increase of 28.51%. The income from DOA (Drug of Abuse) testing achieved 139 million yuan, a year-on-year increase of 15.98%.
Regarding infectious disease testing, COVID-19 antigen tests distributed to Hongkong and mainland of China are main factors for the increase. Furthermore, the increasingly robust influenza in Southern area of China fuels the rehabilitation of flu testing. The HIV-HCV-TP-HBsAg panel remains as a major force to undermine the second curve of regular infectious disease testing domestically. In the public procurement market of WHO, Wondfo has obtained PQ certification for HIV self-test and EUL certification for 2019-nCoV antigen test kit, laying a good foundation for public procurement.
Regarding testing for chronic disease management in the first half year, Wondfo remains devoted to offering multi-dimensional solutions in various scenarios for managing cardiovascular diseases, coagulation diseases, acute respiratory diseases and many other acute and severe illnesses. Revenue from Chemiluminescence Immunoassay, testing of regular items and featured thrombosis testing view considerable increase despite impacts of pandemic control in the overall business pace in the domestic market. In overseas market, revenue from coronavirus-related items witness moderate declining while items for chronic disease management register fast increase. New platforms including chemiluminescence immunoassay and molecular diagnosis have been actively introduced. The company actively promotes the introduction of new platforms such as chemiluminescence and molecular diagnosis. Pilot promotion and establishment of benchmark terminals of new products have been set up to influence neighboring countries, expand overseas business and innovatively expands into various scenarios such at hospital, pediatric, pharmacy, etc. Through these solutions, we strive for sound brand system. Thus, we ensure a sound and sustained development of overseas business.
More Efforts in R&D and Innovation
for Faster Development and Launch of New Products
In recent years, Wondfo Biotech has continuously increased investment in R&D and innovation. In the first half of 2022, the company invested 250 million yuan in R&D, a year-on-year increase of 17.14%. These inputs came out to be 79 new products being developed.
Wondfo Biotech's reserves of cutting-edge technologies have laid a solid foundation for products, and have made breakthroughs in several strategic areas. The Accre System—chemiluminescence immunoassay system of single-cartridge reagent—was exhibited offline in multiple exhibitions held in various countries. The system integrates the excellence of chemiluminescence immunoassay and the flexibility of POCT, which can meet the requirements of primary care for turnaround time and accuracy. At present, Wondfo has further enlarged the product catalog and foster growth prospects of the chemiluminescence platform with an effective synergy in tube type chemiluminescence analyzer and single-cartridge CLIA analyzers.
In the field of molecular diagnostics, Wondfo quickly becomes a major player in the molecular diagnosis track with products related to the nucleic acid detection of COVID-19 (virus sampling tubes, nucleic acid extraction reagents, extractors, and PCR instruments). In the first half of this year, Wondfo’s self-developed U-Box Dx™ Automated Rapid Molecular Diagnostic Instrument obtained approval. U-Box Dx™ automatically finishes the whole detection process including nucleic acid extraction, amplification and analysis, and delivers results within 1 hour with printed report. It can be widely employed to detect pathogens, tumor-related gene mutations, drug-resistance gene mutations, etc.
Regarding pathology business, Wondfo has moved forward from exploration stage to implementation stage. The fully automatic immunohistochemical staining machine PA3600 with independent intellectual property rights has been trialed in many benchmark hospitals nationwide, laying a solid foundation for steady revenue from reagent next year.
At present, the in vitro diagnostic industry is in a stage of rapid development. With “from China, for the World” kept in mind, Wondfo Biotech will carry out strategic layout and business development. We strive to drive sustained and rapid growth in performance through the expansion of endogenous business, and introduce technologies and channel layout through extensional mergers and acquisitions to achieve sustainable development of business portfolio and leading advantages. We continue to improve operating efficiency and consolidate the company's competitiveness in the in vitro diagnostic industry.
 The first developed technology platform with various application scenarios, including infectious disease, fertility, DOA, etc.
The first developed technology platform with various application scenarios, including infectious disease, fertility, DOA, etc. 50+ kinds of reagents and five high-performance devices, focusing on detecting cardiovascular disease, inflammation, kidney injury, sex hormones, thyroid function, diabetes, tumor, and others.
50+ kinds of reagents and five high-performance devices, focusing on detecting cardiovascular disease, inflammation, kidney injury, sex hormones, thyroid function, diabetes, tumor, and others. Single-dose Chemiluminescense Immunoassay Platform
Single-dose Chemiluminescense Immunoassay Platform Wondfo optical blood coagulation analyzer is the first one in the world that can test PT, APTT, TT, FIB, and ACT simultaneously.
Wondfo optical blood coagulation analyzer is the first one in the world that can test PT, APTT, TT, FIB, and ACT simultaneously.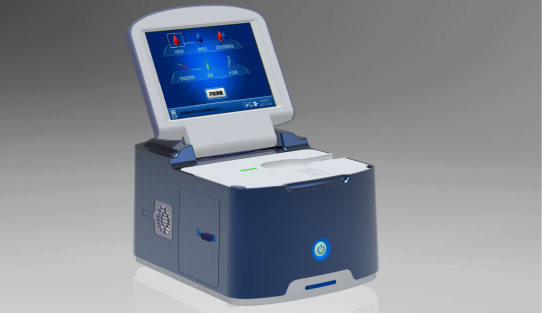 Our Blood Gas Analyzer BGA-102 can produce the result in 30s. Its advantages of portability, easy operation, durability, and high performance make it ideal for clinics, laboratories, and hospitals.
Our Blood Gas Analyzer BGA-102 can produce the result in 30s. Its advantages of portability, easy operation, durability, and high performance make it ideal for clinics, laboratories, and hospitals. Ready-to-use lyophilized RT-PCR Reagent;
Ready-to-use lyophilized RT-PCR Reagent; Wondfo PA-3600 IHC Staining System
Wondfo PA-3600 IHC Staining System The Wondfo Truth-H80E HPLC Hemoglobin Analyzer is a high-performance diagnostic device designed for rapid and precise measurement of glycosylated hemoglobin (HbA1c)—the gold standard for diabetes diagnosis.It support the standard mode and variant mode and provide the result within 60-90seconds.
The Wondfo Truth-H80E HPLC Hemoglobin Analyzer is a high-performance diagnostic device designed for rapid and precise measurement of glycosylated hemoglobin (HbA1c)—the gold standard for diabetes diagnosis.It support the standard mode and variant mode and provide the result within 60-90seconds. Finecare™ Vitamin D, from complexity to simplicity
Finecare™ Vitamin D, from complexity to simplicity Building A World Free from Antibitoic Overuse
Building A World Free from Antibitoic Overuse Advanced rapid diagnostic test with WHO prequalification for infectious disease
Advanced rapid diagnostic test with WHO prequalification for infectious disease The Future Intelligent Medical Assistant to Healthcare
The Future Intelligent Medical Assistant to Healthcare Fight against the pandemic through continuous innovation
Fight against the pandemic through continuous innovation