During November 13-16, MEDICA 2023, the bellwether for the development of the global healthcare industry, was held in Germany. More than 6,000 exhibitors from more than 140 countries and regions showcased cutting-edge technologies and innovative products in the medical industry. With newly launched products, Wondfo made an impression with various products from multiple platforms, manifesting the power of China's intelligent manufacturing and gaining recognition from the audience.
Cutting-Edge Electrochemistry Analyzer
Made Its Debut in MEDICA
As a brand-new cutting-edge analyzer launched from the electrochemistry platform, Ucare-6000, a fully automated blood gas analyzer made its debut at MEDICA. With the features of high efficiency, flexibility, precision and simplicity, Ucare-6000 attracted many professionals and gained appreciation.
Based on the cutting-edge technologies of electrochemical sensing and microfluidic control of the electrochemistry platform, Ucare-6000 adopts the microelectrode film technology, which makes the instrument free of liquid path and easy to maintain. Ucare-6000 is also equipped with a solid quality control system, which can automatically perform calibrations and internal quality control, thus ensuring the accuracy of the results.

"Room-Temperature stability, as a major advantage, can save the trouble of long-distance cold chain transportation and storage, which is very convenient." A customer to our booth expressed his appreciation. Ucare-6000 integrates test card, reagent and QC into one solution pack, which simplifies the manual operation. The reagent pack has a very long validity period at room temperature (2℃-30℃) with a validity of up to 7 months, and it is stable for up to 30 days since its loading.
Ucare-6000 has a comprehensive test menu of 24 calculated parameters including blood gas, electrolytes, metabolites, hematocrit, etc. Furthermore, it can be extended according to the clinical needs to increase specific items such as urea nitrogen and creatinine, which can effectively assist in the clinical and scientific diagnosis.
Various Products from Multiple Platforms
Manifesting China’s Strength in Intelligent Manufacturing
During MEDICA 2023, Wondfo Fluorescence Immunoassay Quantitative System, Mono-Test Chemiluminescence Immunoassay System, Automated Rapid Molecular Diagnostic Instrument, Colloidal Gold Rapid Tests and many products impressed the audience with the strengths of various products from multiple platforms and China's intelligent manufacturing.

During the exhibition, the rapid tests launched in colloidal gold platform was given a lot of attention. Wondfo showcased Rapid Tests of Strep A (Group A Streptococcus) and iFOB, Flu A & Flu B, etc. Based on the colloidal gold immunochromatography method, Wondfo rapid tests are easy to operate with no need for instrument, and the results can be easily interpreted within 10-15 minutes.

The Accre CLIA System with mono-test reagent cartridge, which has been recognized in many international exhibitions, remained to be the focus of this exhibition. The system integrates the sensitivity of chemiluminescence immunoassay and the flexibility of point-of-care testing. It features rapid test and accurate result, convenient operation, and has no liquid path inside, which is only 30% of the size of the traditional CLIA analyzer. The compatible reagent is designed as mono-test reagent cartridge, which are ready-to-use, economical and flexible.

Wondfo started our overseas journey in 2002, bringing China's intelligent manufacturing and Chinese brands to the world. We constantly contribute China's efforts to global health, safeguarding health of everyone and striving to make biology technology benefit the public.
 The first developed technology platform with various application scenarios, including infectious disease, fertility, DOA, etc.
The first developed technology platform with various application scenarios, including infectious disease, fertility, DOA, etc. 50+ kinds of reagents and five high-performance devices, focusing on detecting cardiovascular disease, inflammation, kidney injury, sex hormones, thyroid function, diabetes, tumor, and others.
50+ kinds of reagents and five high-performance devices, focusing on detecting cardiovascular disease, inflammation, kidney injury, sex hormones, thyroid function, diabetes, tumor, and others. Single-dose Chemiluminescense Immunoassay Platform
Single-dose Chemiluminescense Immunoassay Platform Wondfo optical blood coagulation analyzer is the first one in the world that can test PT, APTT, TT, FIB, and ACT simultaneously.
Wondfo optical blood coagulation analyzer is the first one in the world that can test PT, APTT, TT, FIB, and ACT simultaneously.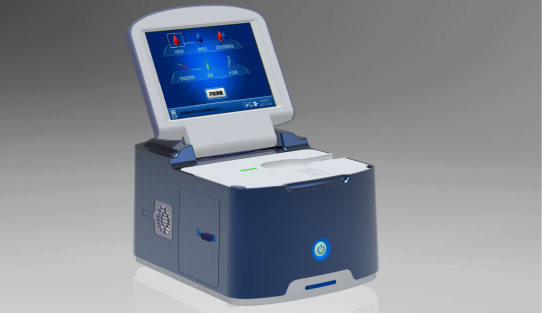 Our Blood Gas Analyzer BGA-102 can produce the result in 30s. Its advantages of portability, easy operation, durability, and high performance make it ideal for clinics, laboratories, and hospitals.
Our Blood Gas Analyzer BGA-102 can produce the result in 30s. Its advantages of portability, easy operation, durability, and high performance make it ideal for clinics, laboratories, and hospitals. Ready-to-use lyophilized RT-PCR Reagent;
Ready-to-use lyophilized RT-PCR Reagent; Wondfo PA-3600 IHC Staining System
Wondfo PA-3600 IHC Staining System The Wondfo Truth-H80E HPLC Hemoglobin Analyzer is a high-performance diagnostic device designed for rapid and precise measurement of glycosylated hemoglobin (HbA1c)—the gold standard for diabetes diagnosis.It support the standard mode and variant mode and provide the result within 60-90seconds.
The Wondfo Truth-H80E HPLC Hemoglobin Analyzer is a high-performance diagnostic device designed for rapid and precise measurement of glycosylated hemoglobin (HbA1c)—the gold standard for diabetes diagnosis.It support the standard mode and variant mode and provide the result within 60-90seconds. Finecare™ Vitamin D, from complexity to simplicity
Finecare™ Vitamin D, from complexity to simplicity Building A World Free from Antibitoic Overuse
Building A World Free from Antibitoic Overuse Advanced rapid diagnostic test with WHO prequalification for infectious disease
Advanced rapid diagnostic test with WHO prequalification for infectious disease The Future Intelligent Medical Assistant to Healthcare
The Future Intelligent Medical Assistant to Healthcare Fight against the pandemic through continuous innovation
Fight against the pandemic through continuous innovation